We’re deep into teaching a Lean LaunchPad class for Life Sciences and Health Care (therapeutics, diagnostics, devices and digital health) at UCSF with a team of veteran venture capitalists. (The class has talked to 1,440 customers to date.)
One of the objectives of the class was to become the Life Science Center of Excellence for the National Science Foundation Innovation Corps. This meant capturing domain specific commercialization expertise for therapeutics, diagnostics, devices and digital health so others can teach this.
Part 1 of this post described the issues in the therapeutics drug discovery pipeline. Part 2 covered medical devices and digital health. Part 3 describedwhat we’re going to do about it. Part 4 gave a snapshot of what one our teams found the first week outside the building.
This is an update of our progress.
It Takes A Village
We’re teaching 110 students in 28 teams across therapeutics, diagnostics, devices and digital health. Teams are made up of clinicians, researchers, and post docs, (some of the team members include the Chief of UCSF General Surgery, the inventor of Fetal Surgery, etc.)
Each of the four cohorts is taught by an experienced life science venture capitalist.Alan May for devices, Karl Handelsman for therapeutics Abhas Gupta for digital health and Todd Morrill for diagnostics.
Jerry Engel and Jim Hornthal, both who taught the National Science Foundation I-Corps classes, are the senior instructors. The UCSF Office of Innovation and Technology (Erik Lium and Stephanie Marrus) is the reason the program exists.
Each of the teams is assigned a mentor to match their domainv(Head of device R&D of Phillips, Genetech, Crescendo, CTO of UCSF, venture capitalists from Sofinnova, Burrill, Lightstone, M34, etc.)
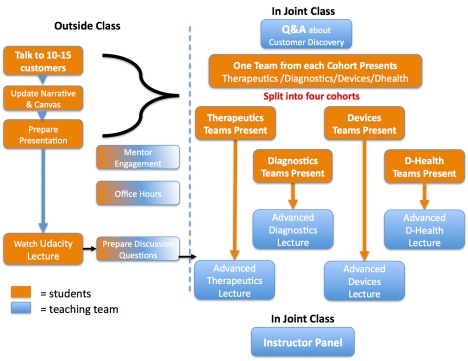
Class Organization – Lots of Moving Parts
Our class meets weekly. We first meet as one group and then we break out the therapeutics, diagnostics, devices and digital health into their own cohorts. The teams present what they learned talking to 10-15 customer/week, and get comments, suggestions and critiques from their teaching team. The instructor then presents a cohort specific lecture explaining how the business model for their area (therapeutics, diagnostics, devices and digital health) builds on and/or differs from the canonical business model in the online Udacity lectures (which the students had to watch as homework.)
We get back together as one group and the instructors share what they learned as they compare and contrast the differences between therapeutics, diagnostics, devices and digital health. We’ve recorded these panels for each part of the business model canvas.
The framework of the class looks like this:
Life Science/Health Care is not a Single Category
One of the reasons I teach is because of how much I learn. One of early surprises of this class for me is finding out that the broad category of “Life Sciences” fails to provide the important nuances of each category to entrepreneurs, investors, educators, policy makers, etc. It turns out that for commercialization, the business model (Customers, Channel, Revenue Model, etc.) for therapeutics, diagnostics, devices, bioinformatics and digital health have very little in common.
You can get a feel for how different by looking at the first two weeks of lectures – covering value proposition and customer segment – from each of the therapeutics, diagnostics, devices and digital health cohorts.
Then at the end of the lectures you can see a “compare and contrast” video and a summary of the differences.
Diagnostics
Week 1 Todd Morrill Instructor
If you can’t see the presentation above click here
Week 2 Todd Morrill Instructor
If you can’t see the presentation above click here
Digital Health
Week 1 Abhas Gupta Instructor
If you can’t see the presentation above click here
Week 2 Abhas Gupta Instructor
If you can’t see the presentation above click here
Devices
Week 1 Allan May Instructor
If you can’t see the presentation above click here
Week 2 Allan May Instructor
If you can’t see the presentation above click here
Therapeutics
Week 1 Karl Handelsman Instructor
If you can’t see the presentation above click here
Week 2 Karl Handelsman Instructor
If you can’t see the presentation above click here
Life Science and Health Care Differences
Once we realized that the four cohorts of therapeutics, diagnostics, devices and digital health were so different we decided to have the instructors compare and contrast howthey’re different for each part of the business model. We’ll be posting these “compare and contrast” videos for every week of the class.
If you can’t see the video above click here
Therapeutics (Starting at 0:30)
- Therapeutics customer = pharma and biotech companies
- Therapeutics Pain & gain = be better than what these companies have in their own drug development pipeline
- Therapeutics Validation = 18 months to a first deal with a potential customer – well before FDA trials, and even before preclinical stage
Digital Health (Starting at 2:40)
- Digital Health Customer = typically consumer end users
- Digital Health Pain & gain = product/market fit needs to be a need, and the value proposition must address it. ”Nice to have’s” do not equal a customer need.
- Digital Health Validation = large scale adoption
Medical Devices (Starting at 6:00)
- Medical Device customers = short term: physicians in private practice and hospital, long term: medical device companies
- Medical Device Customer Goal – figure out the Minimal Viable Product. No such thing as a perfect first generation product that targets a specific physician/customer segment.
- Medical Device Validation= 95% of device startups are acquired by a medical device company, 5% build a large standalone company
Diagnostics (Starting at 10:45)
- Diagnostic Customers = short term In vitro diagnostics are sold to a hospital laboratory or standalone lab, long-term you’ll be bought by Abbott, Roche, etc.
Lessons Learned
- Each of these Life Science domains has a unique business model
- Commercialization of therapeutics, diagnostics, devices and digital health all require the Principal Investigators / founders outside their building talking to customers, partners, regulators
- Only the Principal Investigators / founders have the authority and insight to pivot when their hypotheses are incorrect
- The Lean Startup process and the Lean LaunchPad class can save yearsin commercialization in these domains
- This can be taught