Few spaces were as energized by the pandemic, and the rapid rise of digital health, as decentralized clinical trials, which went from a fairly niche space to a slew of companies suddenly seeing investor interest over the last couple of years.
Perhaps no company within that space benefitted more than Medable, a startup that digitizes clinical trials so that they can be conducted anywhere. On Tuesday, it announced its fourth funding round in the past 18 months, by far its biggest yet.
The $304 million Series D round was co-led by two new investors, Blackstone Growth and Tiger Global, and existing investor GSR Ventures. The round also included follow-on investment from existing investors Sapphire Ventures and WTI. This latest round follows a $78 million round of funding in April, as well as a $25 million funding round and a $91 million round last year, and it brings its total raised a total of $521 million. The company is now is valued at $2.1 billion.
“Our investors understand that Medable is at the forefront of a significant paradigm shift in how clinical research is conducted, which is improving access to clinical trials for populations around the globe. Their investment validates Medable’s fast growth and market presence as the leader in an industry shift to decentralized clinical trials,” Dr. Michelle Longmire, CEO and co-founder of Medable, told VatorNews.
“The faith shown by our investors reflects their belief in Medable’s vision, our team, our strategy, and market opportunity. They support continued technological advancements and expansion that will help Medable’s mission to bring critical new therapies to patients faster than ever before possible.”
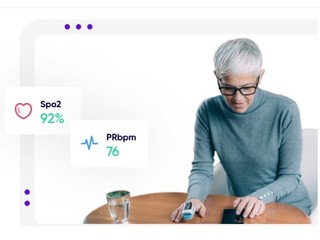
Founded in 2015, Medable’s platform offers its clients, which include clinical trial sponsors, such as pharma and biotech companies, as well as clinical research organizations who manage trials for those companies, features such as patient identification and site selection through AI and machine learning; digitized patient enrollment; and the ability to do trials in any country and in any language.
The company also also provides them with remote patient monitoring, and the ability to see streaming data and patient data in real-time.
Since the start of the pandemic, Medable has been seeing major growth: it onboarded more than 50 new clients in 2020, and grew revenues by more than 400 percent. Its has now deployed its platform via more than 150 decentralized and hybrid clinical trials in 60 countries, serving more than one million patients and research participants globally.
Medable says it will use the funding to expand its team and resources; enhance its platform, product, and operations; and deepen its investment in partner, customer, and patient success.
Part of that means building out its remote patient technologies and tools, as well as to invest more in its SaaS technology platform and partner ecosystem. It also means expanding geographically; this summer, the company announced the first stage of its European expansion plans with a new EMEA headquarters in Dublin, Ireland, which will serve as a hub for expanded sales, customer success, and software development in Europe. The company is also are also planning an expansion into the Asia Pacific region.
“Our vision for the future of clinical research is based on consumer-scale technology that serves billions of patients through mobile applications and connected devices. We look forward to working with more partners, customers, and patients across the globe as we expand our ecosystem and investments,” said Longmire.
“This new funding will help us accelerate global adoption of digital and decentralized clinical trials. It’s a major step forward in our mission to expand clinical research access to every body and every biology, bringing effective therapies to patients faster.”
The decentralized trial space
Among those companies in the decentralized trial space that have raised funding include ObvioHealth, Castor, Antidote, HumanFirst, and Hawthorne Effect. The space even saw one company enter the public market, with Science 37 debuting on Nasqas earlier this month via a SPAC merger.
“The pandemic has accelerated a trend toward decentralized trials that were already underway before it began. Patients want to have the choice of whether to go to a trial site or participate in studies remotely, and that will continue after the pandemic is over,” said Longmire.
“By minimizing the need for in-person site visits, Medable customers have achieved unprecedented results – including 200 percent faster enrollment, 90 percent retention rates, and 50 percent cost reductions.”
As for where she sees the space going next, and where Medable will fit into that ecosystem, she told me that there’s a critical need to expand clinical trial access, improve the efficiency and quality of research, and dramatically improve the patient experience and outcomes. And that is what Longmire wants the company to be able to deliver.
“The future is global adoption of digital and decentralized clinical trials. By working at the intersection of speed, safety and science, we believe Medable can meaningfully reduce the barriers to drug development – and ultimately, enable more effective therapies for people all over the world.”
(Image source: medable.com)