Digital health news, funding round up in the prior week; June 26, 2023
Aledade raised $260M; Zocdoc partnered with Elation Health; Eli Lilly acquired Dice
Read more...
In a strange way, healthcare seems to be reverting back to a style that had gone out of fashion many years ago, with an increased amount of health taking place in the home. There are now around 12 million people who are now getting in-home care, from more than 33,000 providers, and last year the annual expenditures for home health care were projected to be over $72 billion.
This is thanks, in large part, to technology and, more specifically, to connected in-home devices that can easily collect and send data to a physicians in real-time. This allows patients to be monitored remotely, without constant trips to the doctor, and for physicians to do more timely interventions based on patterns picked up by AI and machine learning.
In June, Vator, HP and UCSF Health Hub will be holding an event centered around these devices, and how they are affecting the healthtech space. Every week until then we will be doing a roundup of some of the news around in-home devices and what some of the major tech companies are up to in this space.

Manufacturing solutions provider Jabil consolidated its entire healthcare organization under Jabil Healthcare, to be led by Steve Borges, who will act as CEO and EVP of the division. Jabil Healthcare include Jabil’s embedded-sensors business, its injection molding business through Nypro, and its orthopedics, spine, trauma, and medical instruments operations acquired from Johnson & Johnson.
Jabil Healthcare serves five sectors in the healthcare market: medical devices, orthopedics, diagnostics, pharmaceutical delivery systems, and consumer health.
“By harnessing the power of the Jabil and Nypro teams, we are in the unique position to accelerate technology advancements and augment the value we bring to our customers, their patients and the healthcare industry,” Borges said in a statement. “With the rise of digital health and additive manufacturing, the time is right to unite our diverse capabilities and domain expertise under one strong brand.”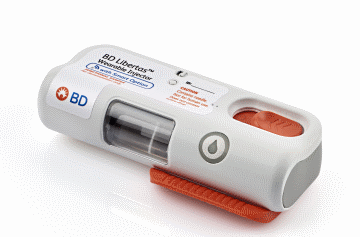 BD finishes clinical trials for wearable drug injector
BD finishes clinical trials for wearable drug injector
Medical device company BD (Becton, Dickinson and Company), a leading global medical technology company, completed a 50-subject human clinical trial with the BD Libertas Wearable Injector, which acts as a drug delivery system that is applied under the skin.
The BD independently sponsored and conducted study was designed to evaluate the performance of the 5 mL BD Libertas device in human subjects, including tissue effects, skin reactivity and patient acceptance. The results are expected to be announced early this year,
“BD is committed to bringing value to our pharma partnerships, including providing them with independent BD sponsored and generated study data to accelerate combination product development. The recent study reflects BD’s continued investment in solutions to meet pharma’s needs by expanding the design space for biologics delivery,” Peter Nolan, Worldwide President, BD Pharmaceutical Systems, said in a statement.
Belgian medical device company Nyxoah, which is focused on the development and commercialization of a hypoglossal nerve stimulation (HGNS) therapy, raised €25 million in a private financing round that included medical equipment company ResMed.
Completion of the financing round is subject to closing conditions and is expected to occur later this month. The company has now raised a total of €66 million.
Nyxoah’s platform is based on the Genio system, a user-centered therapy for OSA, the world’s most common sleep disorder, affecting almost one billion people globally. The funding will go toward developing long-term clinical evidence on the Genio system, prepare for the IDE pivotal trial in the United States and to expand the company's activities in Europe, Australia and New Zealand.
“We have an innovative approach to treating OSA. Having ResMed, the global leader in sleep apnea, support this significant funding round along with our existing shareholders will help Nyxoah to further accelerate its development,” Olivier Taelman, CEO of Nyxoah, said in a statement.
 Is an Alzheimer's wearable on the horizon?
Is an Alzheimer's wearable on the horizon?
Alzheimer’s Research UK launched a global initiative to revolutionise the early detection of neurodegenerative diseases like Alzheimer’s called Early Detection of Neurodegenerative diseases (EDoN). It will harness and analyze digital data to develop signatures of disease – or “fingerprints” – that can be then detected using wearable technologies, such as smart watches.
The goal is to secure at least £67 million over the next six years, and then up to £100 million by 2030, to build and trial its diagnostic device on a large scale. Initial funds have already been secured from Bill Gates and Iceland Foods Charitable Foundation.
Alzheimer’s Research UK will join forces with organisations in data science, clinical and neurodegenerative research to collect and analyze clinical and digital health data such as sleep, gait and speech patterns, to develop early digital fingerprints of diseases like Alzheimer’s.
“During my time as Prime Minister I witnessed first-hand the devastating impact of dementia on families and made a personal commitment to transform the pace of research efforts across the globe. At the first G8 Dementia Summit in 2013, health leaders committed to finding a disease-modifying treatment for dementia by 2025. As that deadline grows closer, we must focus not only on developing life-changing medicines but how we ensure they reach the right people at the right time,” David Cameron, President of Alzheimer’s Research UK, said in a statement.
“Early and accurate diagnosis will give future treatments the head-start they need to succeed. EDoN brings together those at the forefront of this early diagnosis movement. Through technology and big data, I strongly believe we are on the cusp of a revolution in how we detect the brain diseases that cause dementia and radically improve the lives of the millions on families facing these heartbreaking diseases.”
(Image source: mobihealthnews.com)
Aledade raised $260M; Zocdoc partnered with Elation Health; Eli Lilly acquired Dice
Read more...Simple HealthKit and Walmart partnered; Electronic Caregiver raised $95M; Aya bought Flexwise Health
Read more...Alkeus Pharmaceuticals raised $150M; Care.ai partnered with Samsung; DoseSpot bought pVerify
Read more...