Ezra is a direct-to-consumer cancer screening platform that uses a combination of MRI screenings and artificial intelligence to do more accurate cancer screenings, with the goal of making cancer screenings more widely available and more affordable.
The company took one big step closer to that on Tuesday when it announced that it received FDA 510(k) clearance for its AI designed to assist radiologists in analyzing and segmenting prostate MRI. The company also received clearance for its Picture Archiving and Communications System (PACS), called Plexo, which allows radiologists to use the Ezra platform without needing to install any software.
In addition, Ezra also revealed that it is dropping the price of the Ezra prostate scan by 15 percent, from $675 to $575.
“The reason this is very exciting is because our prostate AI was created as a workflow efficiency tool that helps radiologists be more efficient, more productive, when they read prostate MRIs. By doing this, it helps us decrease our cost base, which is helping us decrease our prices, because our goal as a company is to make cancer screening super affordable. This is a step in that direction,” Emi Gal, CEO and co-founder of Ezra, told me in an interview.
The reason the AI allows the New York-based company to reduce the price is because, as a result of using the platform, radiologists will be spending about half the time they are currently spending on reading a prostate MRI, he explained.
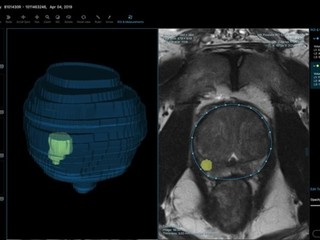
When a radiologist reads a prostate MRI, there are three things they need to do: measure the size and volume of the prostate, because the volume of the prostate is potentially indicative of cancer. They need to segment the prostate, which basically means drawing a border around the prostate across every single MRI slide. And then, if there is a lesion, they need to segment the lesion for biopsy prep.
“All of those segmentations, measurements, volume renderings, our AI does that automatically. So, we’re saving radiologists probably about 10 minutes per study with the AI, which enables us to have lower costs, which leads to lower prices, because really what we want to do is we want to pass all of our cost savings created by the AI to our customers. We’re decreasing the price because of that,” said Gal.
Founded in 2017, Ezra’s platform allows patients to schedule appointments with a RadNet imaging facility; when they arrive, they are greeted by a member of the Ezra staff at the facility who then guides them through the process. The member gets their scan done and then the results are made available within their Ezra interface. They can also use the platform to schedule a call or a meeting to speak to the Ezra physician to be taken through the results.
The company partners with existing outpatient imaging facilities and all Ezra scans are analyzed by board-certified radiologists. The company’s cancer screening programs are currently live in 11 facilities across New York City, San Francisco, and Los Angeles.
In addition to providing prostate scans, Ezra also does lung scans, as well as a full-body MRI scan, which cover 14 internal organs. All scans other than prostate MRIs, however, do not currently use artificial intelligence, as the FDA clearance does not cover those.
“Our offering has two parts: it’s the actual scan, and for the actual scan we use FDA approved MRI sequences. So, like our prostate scan is a typical prostate scan. And then there’s the interpretation of those scans, which generates a radiology report. For the interpretation front, we are currently only using AI for the prostate because that’s what has been cleared,” said Gal.
“For all the others, for full body, for the lung scan, etc. we are building AI that we will go through the process with the FDA to have cleared. So, those scans that you see listed on the website for now do not use AI for the reading, with the exception of prostate.”
So far, Ezra has scanned over 1,000 people since it went live, and has helped 42 people find cancer, which equals roughly 4 percent of its members. That is a much higher percentage than the typical detection rate, which is less than 1 percent.
“The great thing about Ezra is that our scan covers about 14 organs in the body, and for a lot of these organs there are currently no screening guidelines. For example, with the exception of breast, lung, colon, prostate and uterine cancer, none of the other cancers have a way to screen. So, if you’re talking about brain cancer, liver, gallbladder, pancreas or kidney cancer, you’ll only find cancer in those organs if you’re symptomatic. And if you’re symptomatic, it’s generally too late,” Gal explained.
Without Ezra, he said, you could not even find cancer in these organs.
“So, the great thing about doing an Ezra scan is you screen for cancer in all of those organs, which then enables you to find problems before you’re symptomatic, which means that it’s more likely to be an early stage cancer. Early stage cancer is super important to detect because the five year survival rate for stage 1 cancer is about 80 to 90 percent, whereas the five year survival rate for stage 4, or late stage, cancer is less than 20 percent. Early detection leads to better outcomes.”
This FDA clearance will enable the company to roll out its AI across all of its imaging partners and imaging facilities, with the plan going forward to expand to more markets beyond the three it is currently live in.
“I want Ezra to be synonymous with cancer screening, and for that we need to really make cancer screening comprehensive, which we’re doing with our full body. We need to make it affordable, which we’re doing with our AI. We really are focusing on making cancer screening widely available and affordable, and our AI is pointing us us towards that direction,” said Gal.